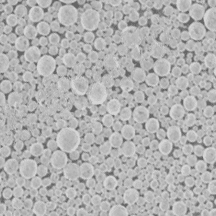
Description
Lithium Titanate or lithium titanium oxide (Li4Ti5O12 Spinel, or LTO) is an electrode material with exceptional electrochemical stability. It is used as the anode material in lithium-ion batteries for applications that require long cycle life, high rate, and high coulombic efficiency.
Lithium titanium oxide has some advantages as an anode material, such as fast charging, long cycle life, and high safety. It can also operate at lower voltages than other anode materials, such as graphite, which means it can avoid the formation of lithium dendrites that can cause short circuits. However, lithium titanium oxide also has some drawbacks, such as low specific energy, high cost, and poor low-temperature performance.
It is usually coupled with a LiFePO4 or NCM transition metal oxide cathode to form a full lithium-ion cell. Although the cell voltage is lower than the graphitic carbon-based cell, LTO-based batteries are considered safer and have a wider operating temperature range.

Specifications
| D50 (um) | 14 |
|---|---|
| BET (m2/g) | 2.5 |
| Tap density (g/cm3) | 1.5 |
| Capacity (mAh/g) | 164 |
| Voltage cut-off (V) | 1.0~2.8V |
| Test condition | 25 Degree C, 0.1C 1.0~2.8V |
Reference: Electrochemical properties of lithium–titanium oxide, modified with Ag–Cu particles, as a negative electrode for lithium-ion batteries – RSC Advances





Reviews
There are no reviews yet.